WHAT IS ACNE?
Acne vulgaris is a chronic dermatologic disease relating to blockage and/or inflammation of the pilosebaceous unit. It can be characterized by non-inflammatory (comedone) and inflammatory (papule and pustule) skin spots which mostly appear in the sebum-rich areas such as face, back, and chest. The pathogenesis of acne is probably multifactorial, it being related to an interaction among some factors such as increased sebum production by pilosebaceous glands, keratinocyte hyperproliferation, bacteria colonization (especially Propionibacterium acnes, i.e. P. acnes) in hair follicle, primary perifollicular inflammation, and inflammatory responses to P. acnes metabolites, cell debris and/or sebum.
Therefore, acnegenicity clinical trials for cosmetic products can be carried out for two purposes: to evaluate the effectiveness of test products in the treatment of acne lesions or to evaluate the acnegenic potential of test products. In both scenarios, we can support some interesting claims based on Expert grader assessment and/or instrumental analysis:
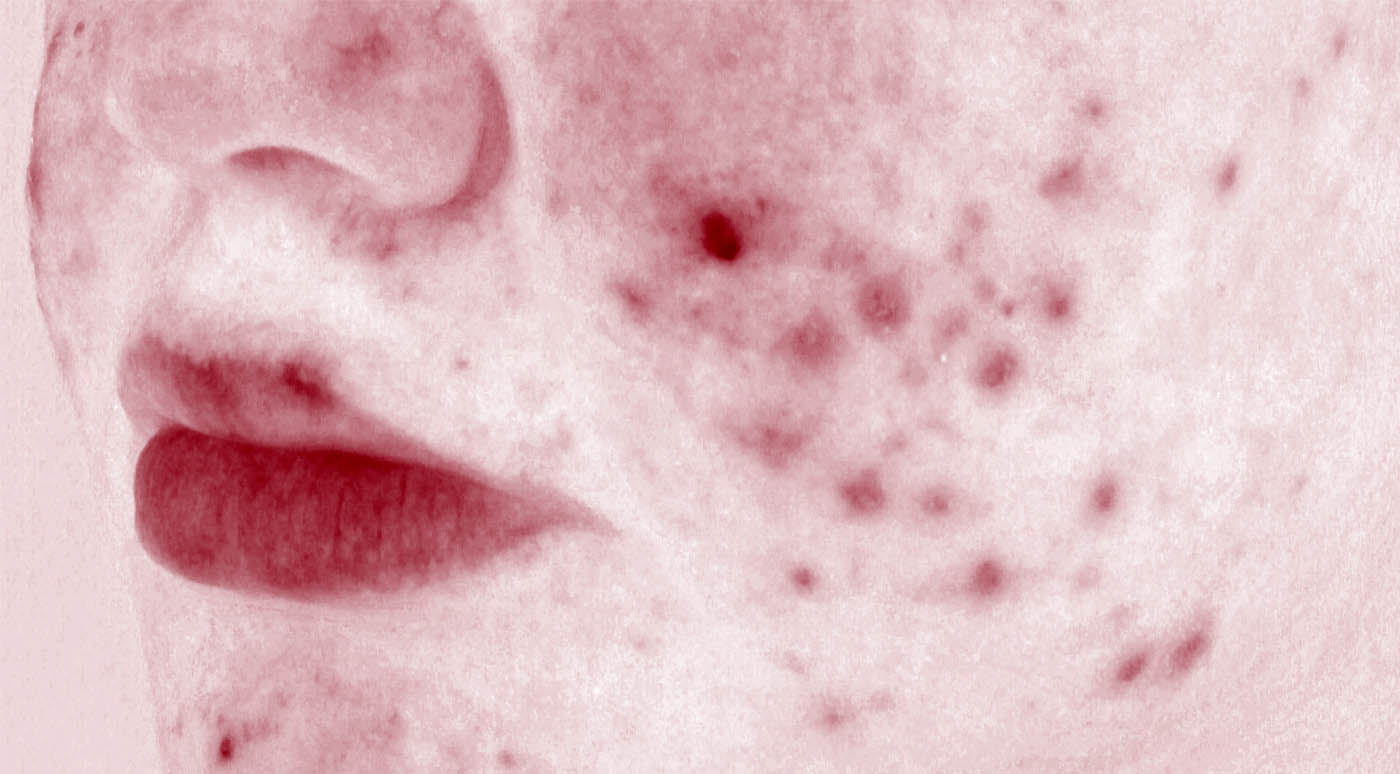
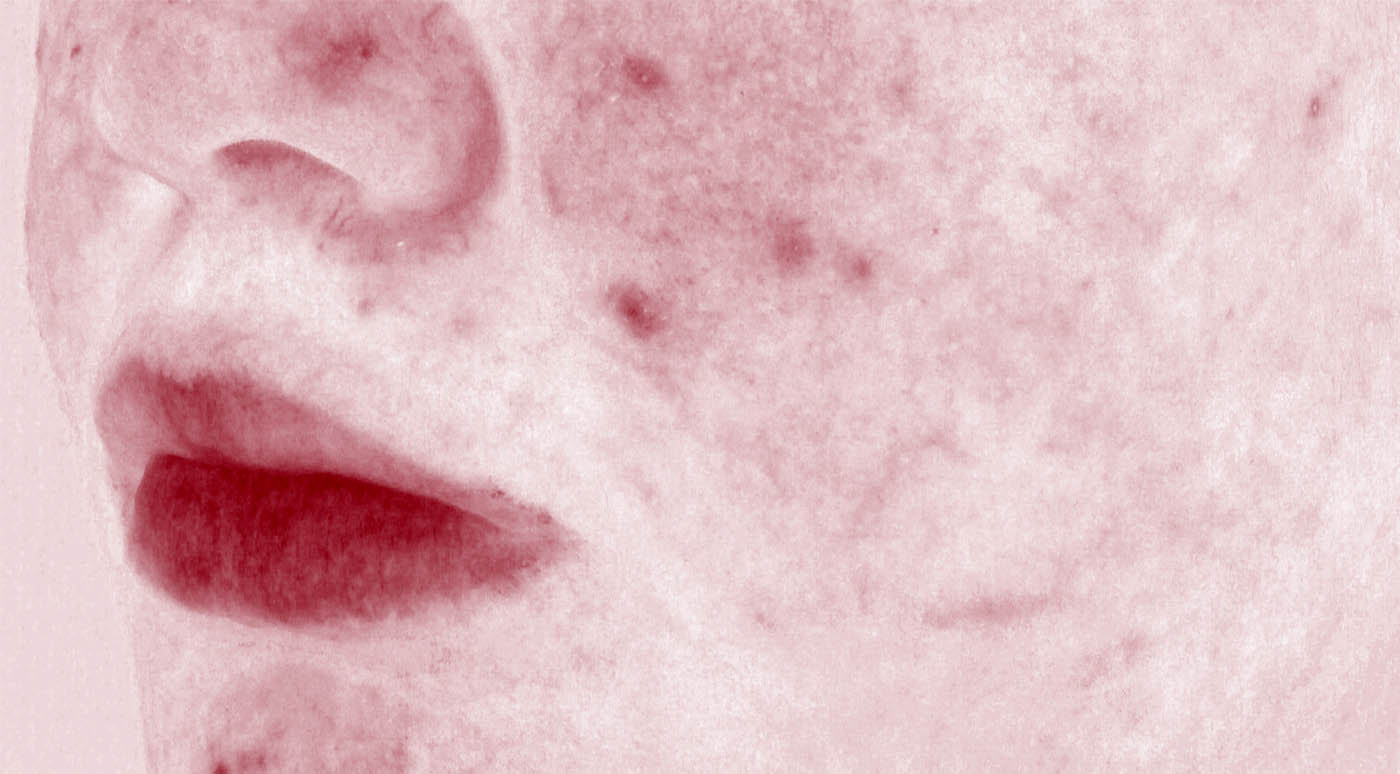

WHAT CLAIMS CAN WE SUPPORT?
- Dermatologically tested
- Clinically Proved
- Antiacne
- Minimize the appearance of pores
- Oil control
- Helps to remove dirt and dead skin cells
- Balances pH
- Hydrating
- Helps boost skin’s self defense
- Reduce inflammation

 Portuguese
Portuguese
 Español
Español
